CVS Pharmacy and Walgreens are no longer selling Zantac, as well as generic version of the heartburn medication, over concerns that they might contain a substance that could cause cancer.
CVS, the largest pharmacy chain in the country, said over the weekend it was suspending sale of Zantac and other products which contain ranitidine until further notice. Walgreens said in a statement it is removing the medications from shelves while the Food and Drug Administration (FDA) continues its review of the products.
The FDA said earlier this month that it has learned that some ranitidine medications—including those known by the brand name Zantac—contain low levels of an impurity that could cause cancer.
Zantac and its CVS-branded generic counterpart have not been recalled, a statement from CVS said, and the FDA has not recommended that patients stop taking ranitidine products. Customers who have already bought the products can return them to CVS for a refund, CVS said.
Other medications under investigation
The nitrosamine impurity known as N-nitrosodimethylamine, or NDMA, has been classified as a probable human carcinogen based on lab tests, and this isn't the first time that it has been detected in a common medication.Since last year, the FDA has been investigating NDMA and other impurities in blood pressure and heart failure medicines known as angiotensin receptor blockers or ARBs. Numerous recalls have been launched as the FDA found "unacceptable levels" of nitrosamines in several of those common drugs containing valsartan.
An Update From the FDA
Some months ago, the FDA released a list of nitrosamine-free blood pressure medications that are safe to use.On April 5, a list of approximately 40 blood pressure and heart medications that don’t contain the cancer-linked nitrosamine, was posted by the agency.
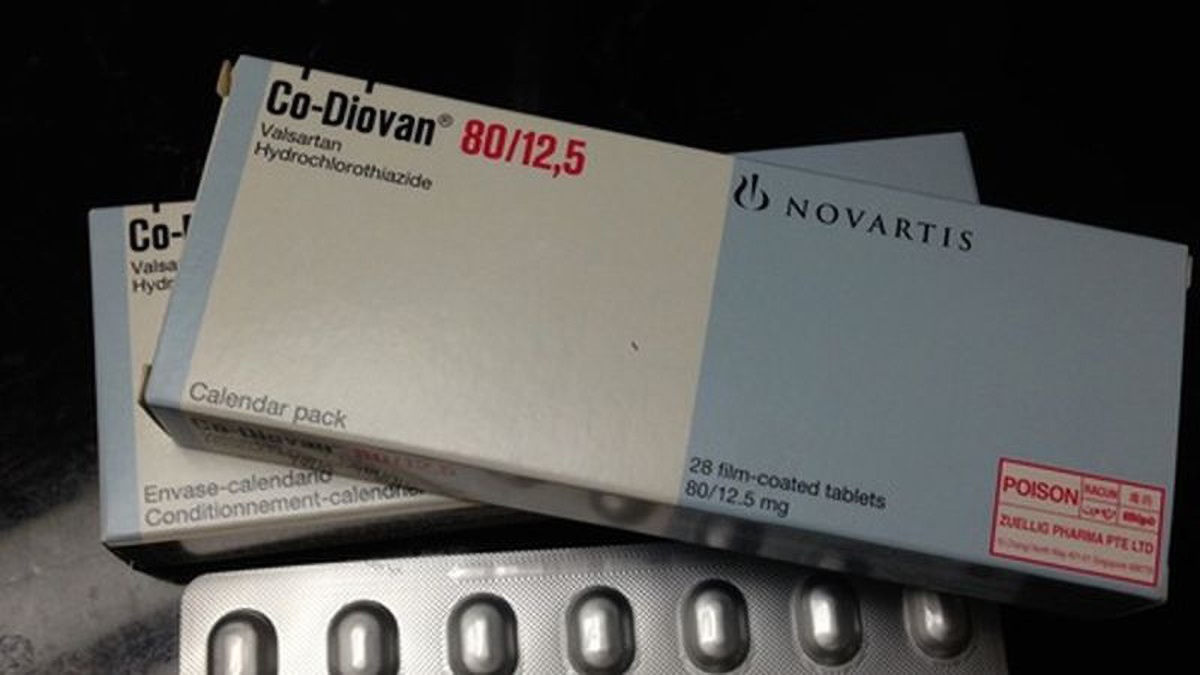
The issue initially surfaced in July of 2018, when the FDA claimed they recalled several medications with valsartan after impurities were discovered. Those recalled medications were said to be manufactured by a drugmaker located in China.
According to an April 4 press release, the FDA has since then “been working to thoroughly understand how this issue arose, how we could address and mitigate exposure to this unnecessary risk to patients, and what we could do to prevent these types of impurities from reoccurring in future medications.”
In the statement, the agency added that the presence of nitrosamines in drug products is “not acceptable.”
“We’re also continuing to work with manufacturers to swiftly remove medications from the market if they contain a nitrosamine impurity at levels higher than the interim acceptable intake limits,” the FDA said.
“Removing the affected medications from the market has led to shortages, and since then, we’ve been working to mitigate and prevent shortages as often as possible.”
N-dimethylnitrosamine (NDMA), N-diethylnitrosamine (NDEA), 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK), are among common nitrosamines.
The list of “safe” medications can be found on the FDA’s website. Drugs marked “not present,” mean the FDA “has completed the comprehensive assessment noted above.”

