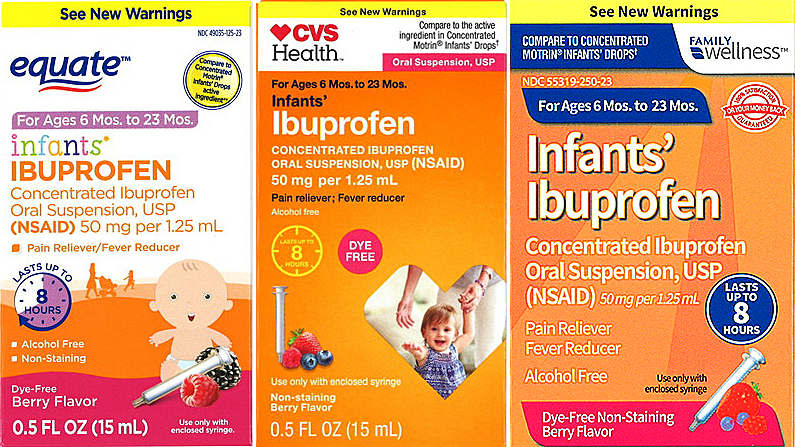
Drug manufacturer Tris Pharma has announced a nationwide recall of infant-dosed ibuprofen sold at CVS, Walmart, and Family Dollar stores.
In the announcement, Tris says that three separate lots of its liquid ibuprofen solution for infants with a slightly higher concentration of the drug than intended got through to retail distributors.
The chief fear is that the higher concentration of ibuprofen could cause kidney damage in infants and young children.
Tris warns that the product, 0.5 oz. bottles of Infants' Ibuprofen Concentrated Oral Suspension, USP (NSAID) 50 mg per 1.25 mL, could also cause “nausea, vomiting, epigastric pain, or more rarely, diarrhea.
“Tinnitus, headache, and gastrointestinal bleeding are also possible adverse effects.”
Tris also pointed out that as of the publication of the recall order, there had been no reports of any infants having bad reactions.
The recall is precautionary.
The extra strong ibuprofen was sold to a single supplier who distributed the product to the three pharmacy chains.
Tris advises wholesalers and retailers who are carrying these products to check lot numbers and immediately remove from their shelves any of the potentially dangerous bottles.
The affected lot numbers are 00717009A with expiry date 02/19, 00717015A with expiry date 04/19, 00717024A with expiry date 08/19, 00717024A with expiry date 08/19, and 00717024A with expiry date 08/19.