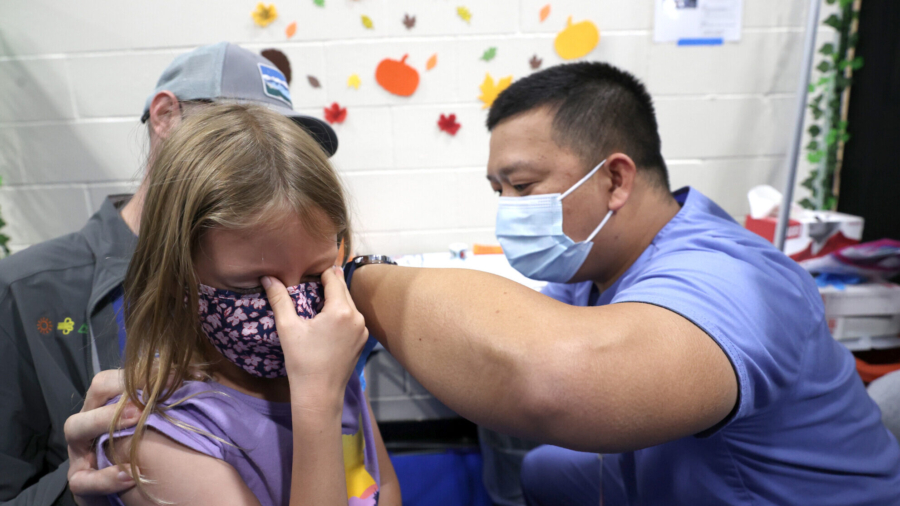
Pfizer and BioNTech on Tuesday asked the U.S. Food and Drug Administration (FDA) to grant emergency use authorization for a booster for their COVID-19 vaccine for children aged 5- to 11-years-old.
Earlier this month, the companies released data from a mid-to-late stage study that they said showed a boost in antibodies from a third dose of their vaccine, providing increased protection against the novel coronavirus and the Omicron variant.
The trial saw 140 children be administered a booster dose approximately six months after the second dose. The shot comes in a two-dose primary series.
The children saw a 6-fold increase in antibodies against the Wuhan strain of SARS-CoV-2 one month after the booster, the companies said. A sub analysis of 30 participants demonstrated a 36-fold increase in neutralizing antibody titers.
“Data from this study demonstrated a strong immune response in this age group following a booster dose of the Pfizer-BioNTech COVID-19 Vaccine with no new safety signals,” the companies said in a statement.
The data has not yet been published on a preprint server or in a journal.
According to data from the Centers for Disease Control and Prevention (CDC), 28 percent of children aged 5- to 11-years-old—around 8.2 million—were fully vaccinated as of April.
There has been some skepticism on the need for boosters in younger children given the reduced risk of severe infection and hospitalization in the age group.
Pfizer and BioNTech have asked the FDA to grant emergency use authorization for a 10-microgram booster dose for children in the age group. Adults are given a 30-microgram dose of the vaccine.
The primary two-dose COVID-19 shot from Pfizer and BioNTech was authorized in the United States for children aged 5- to 11-years-old in October 2021.
The FDA in January authorized the use of a booster dose of the Pfizer and BioNTech vaccine for children aged 12 to 15. The agency has also authorized a booster shot for children aged 5 through 11 years who are immunocompromised.
Pfizer CEO Albert Bourla also said in a podcast interview last week that he hopes the companies’ COVID-19 vaccine will be granted emergency use authorization by the FDA for children under 5 years old in June.
The FDA has said it does not comment on submissions pending before the agency but a spokeswoman told The Epoch Times last month that “our approach has always been to conduct a regulatory review that’s responsive to the urgent public health needs created by the pandemic, while adhering to our rigorous standards for safety and effectiveness.”
Drug companies like Pfizer “do not apply for product distribution approvals from the FDA unless they are reasonably confident the FDA will approve,” Barbara Loe Fisher, co-founder and president of the National Vaccine Information Center, previously told The Epoch Times in an email.
The submission being made based on data from the small number of children is “a new low for scientific evidence of a vaccine’s safety and efficacy and a clear indication that the pharmaceutical industry views FDA as a rubber-stamping agency,” she added.
Zachary Stieber and Reuters contributed to this report.
From The Epoch Times