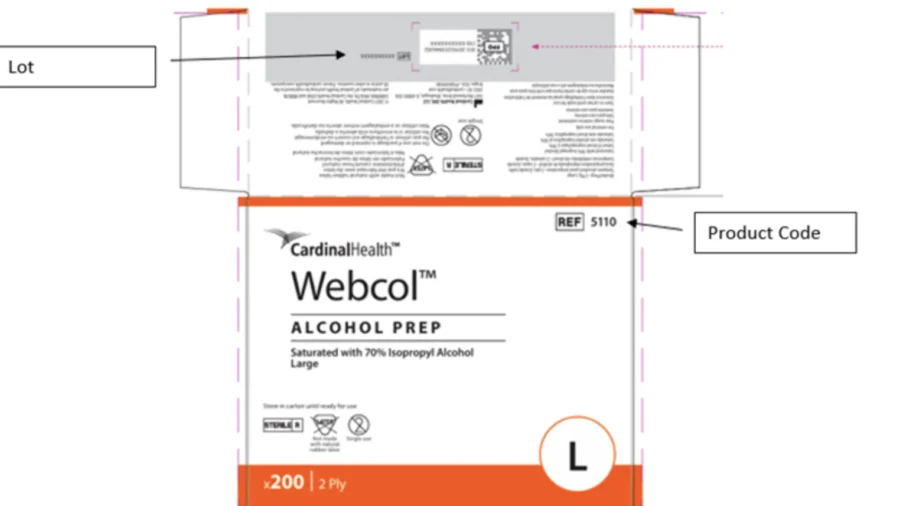
The contaminated alcohol prep pads were sold to both businesses and individual consumers, posing a potential health risk, especially for people with weakened immune systems.
The affected product is the large, 70 percent isopropyl alcohol prep pad, distributed in the United States, Puerto Rico, and Japan from September 2025 to February 2026. The product is used for external skin cleansing.
“There is a reasonable probability that use of the contaminated pads may result in local and systemic infections in at-risk populations,” the recall notice states. “Patients with impaired immune response, including patients with cancer receiving chemotherapy or poorly controlled diabetes, may develop life-threatening infections such as bacteremia or central nervous system infections.”
Cardinal Health alerted business customers to the recall by overnight mail in early March, instructing them to check their inventory, isolate affected products, notify downstream recipients, and confirm receipt of the notice, according to the company and FDA recall notice.
"Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to taking or using this drug product," stated the recall.
Consumers with questions about the recall can also contact Cardinal Health customer service by email or phone, as provided on the recall notice.
When a company announces a recall, market withdrawal, or safety alert, the FDA publishes the notice as a public service.
The FDA also urges consumers to report adverse reactions to its MedWatch program at www.fda.gov/medwatch.