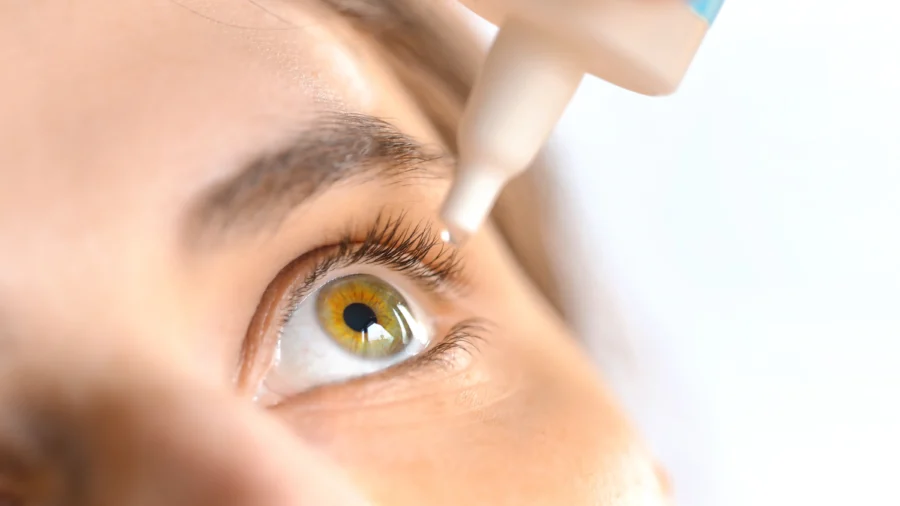
Over 75,000 units of eye care products distributed across the United States have been voluntarily recalled by pharmaceutical consulting firm BRS Analytical Services due to manufacturing violations that could compromise product quality, according to a recall notice.
The recall covers multiple ophthalmic solutions and gels including: Artificial Tears Ophthalmic Solution, Carboxymethylcellulose Sodium Ophthalmic Gel 1 percent, Carboxymethylcellulose Sodium Ophthalmic Solution, Lubricant Eye Drops Solution, and Polyvinyl Alcohol Ophthalmic Solution. These products were shipped between May 26, 2023, and April 21, 2025.
According to the recall notice, the health hazard to users is currently unknown, but cGMP deviations can result in products of unacceptable quality, and it is not possible to rule out patient risks resulting from their use.
Current Good Manufacturing Practices are a set of regulations enforced by agencies such as the FDA to ensure that products are produced consistently and controlled according to quality standards. These guidelines cover every aspect of the manufacturing process, including facility design, equipment calibration, staff training, production controls, and documentation. The goal is to prevent contamination, production errors, and other issues that could lead to unsafe or ineffective products.
According to AvKARE, customers are asked to complete a return form and send any recalled products to R&S Northeast LLC in Fountain Run, Kentucky. Full credit, including shipping costs, will be provided for returned items. The company said it regrets any inconvenience caused by the recall and encouraged anyone with questions to contact their customer service department.
The affected products, sold under the brand names Fresh & Ready Foods, City Point Market Fresh Food to Go, and Fresh Take Crave Away, are marked with “use by” dates from April 22 to May 19, 2025.
“Individuals with an allergy or severe sensitivity to soy risk serious or life-threatening allergic reactions if they consume this product,” the company said.