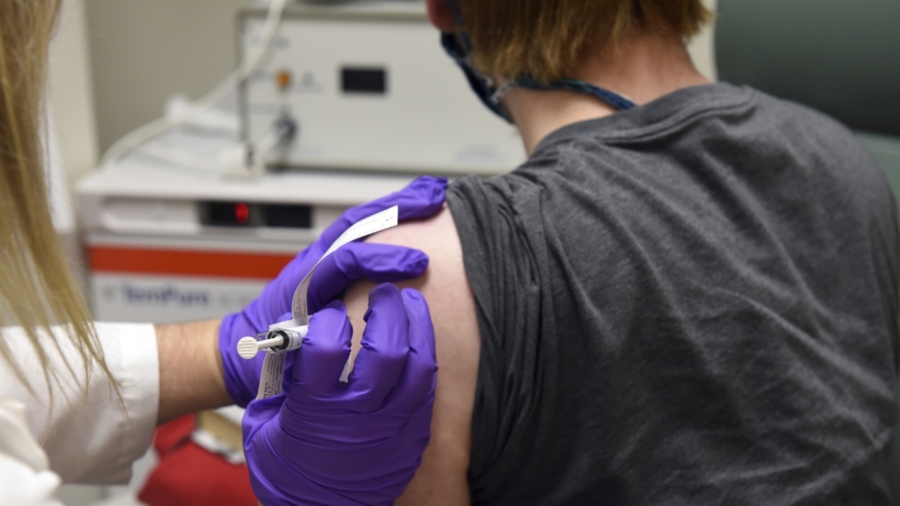
A COVID-19 vaccine candidate proved strongly effective in a large phase 3 study, according to results released on Nov. 9.
The results were analyzed by an independent data monitoring board. They indicate an efficacy rate above 90 percent at seven days after the second dose, New York-based Pfizer and German biotechnology company BioNTech said. That means protection is achieved 28 days after the first vaccine. The vaccination schedule is two doses.
No serious safety concerns were reported in the interim results.
“Today is a great day for science and humanity. The first set of results from our Phase 3 COVID-19 vaccine trial provides the initial evidence of our vaccine’s ability to prevent COVID-19,” Dr. Albert Bourla, Pfizer chairman and CEO, said in a statement.
“The first interim analysis of our global Phase 3 study provides evidence that a vaccine may effectively prevent COVID-19. This is a victory for innovation, science and a global collaborative effort,” added professor Ugur Sahin, BioNTech co-founder and CEO.
The phase 3 trial started on July 27 and has enrolled over 43,000 patients to date.
Nearly 39,000 have received the second dose as of Nov. 8.
There are currently no approved vaccines for the CCP virus. Dozens are in development around the world.
Vice President Mike Pence called the development "HUGE News," adding: "Thanks to the public-private partnership forged by President @realDonaldTrump, @pfizer announced its Coronavirus Vaccine trial is EFFECTIVE, preventing infection in 90% of its volunteers."
"STOCK MARKET UP BIG, VACCINE COMING SOON. REPORT 90% EFFECTIVE. SUCH GREAT NEWS!" Trump added in a tweet.
The U.S. government reached a deal with Pfizer and BioNTech in July, agreeing to pay $1.95 billion for the first 100 million doses of BNT162, the vaccine candidate the two companies created.
The deal was contingent upon approval or emergency use authorization from the Food and Drug Administration, which is still pending.
Democratic presidential nominee Joe Biden issued a statement congratulating "the brilliant women and men who helped produce this breakthrough and to give us such cause for hope."
Dr. Richard Hatchett, CEO of the Coalition for Epidemic Preparedness Innovations (CEPI), which has been helping fund various vaccine candidates, said the results were highly positive.
"We believe these interim results also increase the probability of success of other COVID-19 candidate vaccines which use a similar approach [pre-fusion spike as their immunogen], including all of the vaccines in the CEPI portfolio," he said in a statement.
"If the final longer term analysis of the study data confirms this result, and if no safety issues are identified in the trial participants, this vaccine candidate will be able to seek regulatory approval."